WHAT CAUSES FIBROID FORMATION?
The question most women ask is what causes fibroids? Simply answered: we don’t know because we do not have the complete picture. Some cultures believe some wicked persons cast fibroid as a cause on women or that witches are responsible for fibroid formation. We do know that fibroids occur in some races more frequently than others. African women of child bearing age have a higher incidence of fibroids than their caucasian, Asian or latino women. We also know that fibroids can run in families. If your mother has fibroids, there is an increased chance that you may develop fibroids later in life. We think that fibroids form as a result of many factors among which may include but not limited to genetic, hormonal ( ESTROGEN AND PROGESTORONE RECEPTORS LEVELS ), environmental and other things interracting together. Many African women with fibroids go through life, have their children and die without symptoms or ever knowing they have fibroids.
Many successful pregnancies have occured in women with co-existing fibroids.
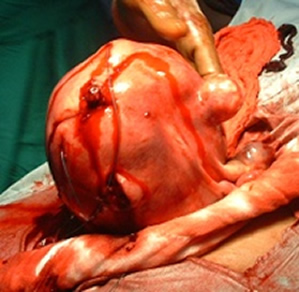
Their sizes vary greatly from tiny ones like palm kernel nuts to others that can be as large as soccer ball.
Most fibroids grow slowly over years. Each fibroid starts its growth by transformation of a single muscle cell of the uterus. Most fibroids grow within the wall of the uterus. These types are called intra-mural fibroids.
Others occur less frequently inside the cavity of the uterus called endometrium .
SUB-MUCOUS FIBROID (SIS) SUB-MUCOUS FIBROID (SIS)
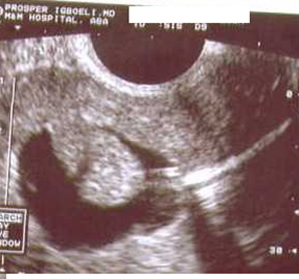 SUB-MUCOUS FIBROID
| 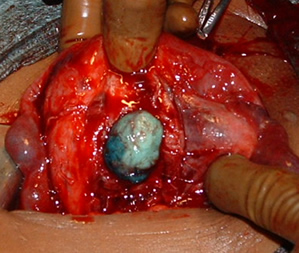 SUB-MUCOUS FIBROID AT SURGERY |
These can cause serious problems and infertility for the woman. These are called sub-mucous fibroids. They occupy space where the fetus will normally develop.
Some fibroids also occur on the surface of the womb and are more harmless. They are called sub-serous or pedunculated fibroids. These do not cause much infertility problems for the women unless they block their tubes or occupy space due to their sizes.
PEDUNCULATED AND SUB-SEROUS FIBROIDS
Fibroids can also occur in the presence of pelvic adhesions and tubal disease and this combination can cause great difficulty at surgery with its attendant morbidity.
BLOCKED TUBE, FIBROIDS AND ADHESIONS (SCAR TISSUE) FROM INFECTION
Many women have fibroids without knowing about it because the fibroids do not cause any problems for them. They do not have symptoms and the fibroids have not prevented them from having babies.
We don't know what causes fibroids to grow or shrink. We do know that they are under some hormonal control — both estrogen and progesterone. They shrink when anti-hormone medications like GNRH ANALOGS are used. They also stop growing or shrink once a woman reaches her menopause. They are very rarely found before menarche or pre-puberty.
Lots of research data are available that are helping us understand fibroids more and more. The studies are carried out mostly in western countries where fibroid prevalence and incidence are low. Listed below are summaries of findings or conclusions from few of such studies.
Fibroids are monoclonal tumors and approximately 40 to 50% show abnormal karyotype. Certain mutations of some proteins have been noted in 70 percent of fibroids.
Exact cause of fibroids is not clearly understood, but the current working hypothesis is that genetic predispositions, prenatal hormone exposure and the effects of hormones, growth factors and xenoestrogens cause fibroid growth.
Known risk factors are Black race, nulliparity, obesity, polycystic ovary syndrome, diabetes and hypertension.
Fibroid growth is strongly dependent on estrogen and progesterone. Although both estrogen and progesterone are usually regarded as growth-promoting they will also cause growth restriction in some circumstances. Pregnancy appears to exert a certain protective effect.
This protective effect might be partially mediated by an interaction between estrogen and the oxytocin receptor.
It is believed that estrogen and progesterone have a mitogenic effect on leiomyoma cells and also act by influencing (directly and indirectly) a large number of growth factors, cytokines and apoptotic factors as well as other hormones. Furthermore, the actions of estrogen and progesterone are modulated by the cross-talk between estrogen, progesterone and prolactin signalling which controls the expression of the respective nuclear receptors. It is believed that estrogen promotes growth by up-regulating IGF-1, EGFR, TGF-beta1, TGF-beta3 and PDGF, and promotes aberrant survival of leiomyoma cells by down-regulating p53, increasing expression of the anti-apoptotic factor PCP4 and antagonizing PPAR-gamma signalling. Progesterone is thought to promote the growth of leiomyoma through up-regulating EGF, TGF-beta1 and TGF-beta3, and promotes survival through up-regulating Bcl-2 expression and down-regulating TNF-alpha. Progesterone is believed to counteract growth by downregulating IGF-1. Expression of transforming growth interacting factor (TGIF) is increased in leiomyoma compared with myometrium. TGIF is a potential repressor of TGF-β pathways in myometrial cells.
Whereas in premenopausal fibroids the ER-beta, ER-alpha and progesterone receptors are found overexpressed, in the rare postmenopausal fibroids only ER-beta was found significantly overexpressed. Most studies found that polymorphisms in ER and PR gene encodings are not correlated with incidence of fibroids in Caucasian populations however a special ER-alpha genotype was found correlated with incidence and size of fibroids. The higher prevalence of this genotype in black women may also explain the high incidence of fibroids in this group.
Uterine leiomyoma was more sensitive than normal myometrium to PPAR-gamma receptor activation resulting in reduced survival and apoptosis of leiomyoma cells. The mechanism is thought to involve negative cross-talk between ER and PPAR signaling pathways. Several PPAR-gamma ligands were considered as potential treatment. PPAR-gamma agonists may also counteract leiomyoma growth by several other mechanisms of action such as TGF-beta3 expression inhibition.
Hypertension is significantly correlated with fibroids. Although a causal relationships is not at all clear the hypothesis has been formulated that atherosclerotic injury to uterine blood vessels and the resulting inflammatory state may play a role. Furthermore endocrine factors related to blood pressure such as angiotensin II are suspected to cause fibroid proliferation via angiotensin II type 1 receptor.
Aromatase and 17beta-hydroxysteroid dehydrogenase are aberrantly expressed in fibroids, indicating that fibroids can convert circulating androstenedione into estradiol. Similar mechanism of action has been elucidated in endometriosis and other endometrial diseases. Aromatase inhibotors are currently considered for treatment, at certain doses they would completely inhibit estrogen production in the fibroid while not largely affecting ovarian production of estrogen (and thus systemic levels of it). Aromatase overexpression is particularly pronounced in black women
Genetic and hereditary causes are being considered and several epidemiologic findings indicate considerable genetic influence especially for early onset cases. First degree relatives have a 2.5-fold risk, and nearly 6-fold risk when considering early onset cases. Monozygotic twins have double concordance rate for hysterectomy compared to dizygotic twins.
Like keloids, fibroids have disregulated production of extracellular matrix. Recent studies suggest that this production may represent an abnormal response to ischemic and mechanical tissue stress. Several factors indicate significant involvement of extracellular signaling pathways such as ERK1 and ERK2, which in fibroids are prominently influenced by hormones. Paradoxically and unlike most other conditions involving significant fibrosis the Cyr61 gene has been found downregulated in fibroids.
Cyr61 is also known for its role as tumor suppressing factor and in angiogenesis. Hence fibroids are one of the very few tumors with reduced vascular density.